Phase 2 at a Glance
|
Phase 2 Medical Manufacturing, Inc., a precision medical device manufacturer, had been pursuing a lean transformation for nearly five years and had a lot to show for it. The company had small lot production, elements of visual management, ongoing training in lean tools for everyone, and an open, respectful culture. But when profit margins took a hit to meet a threat from offshore competition, the company had to get its lean effort to a much higher level – fast.
“It was time to make some big changes and make sure that they would stick,” said Adam Prime, Phase 2 president.
The situation began when main customer Medtronic Advanced Energy (MAE) put out for global bidding the combined volume on single-use medical devices made by Phase 2 in Rochester, NH, and by an MAE facility just 25 miles away in Portsmouth, NH. The disposable medical device is used in various types of surgery.
“You had manufacturing here at Phase 2 that was being duplicated by manufacturing at Medtronic in Portsmouth,” explained Prime. “We were making the same products.”
Duplicate production facilities meant duplicate engineering and management resources. Plus, the core competencies of each facility were sub-optimized. Portsmouth’s forte was new product development while also manufacturing roughly 50% of the volume. Phase 2, the larger production facility, specialized in contract medical device manufacturing, but also did some new product development.
 |
|
A Phase 2-produced device. |
For MAE, combining production and seeking a global price from a single overseas source made sense in the highly competitive medical device manufacturing market. But it would also make Phase 2’s life precarious. The loss of manufacturing MAE’s products would have been a significant negative impact on Phase 2’s revenue.
Losing the MAE work – and the companion jobs – “would have hurt,” said Prime. “It would have been painful but we would have survived.”
His biggest fear was that losing the MAE business would be the first step in losing everything to overseas competition. “Once that door is open, it’s only a matter of time,” he said.
Two Lean Journeys Join
Medtronic solicited bids on the combined volume from device manufacturers in China, India, and Mexico, receiving globally competitive options. But there were compelling reasons for MAE to keep the business at Phase 2. It had deep knowledge of the product and manufacturing processes, making improvements to both over the course of 12 years.
Medtronic, which is on a lean journey and wants suppliers to follow its “Cell Operating System,” weighs more than factory-gate pricing in supply chain decisions. Extending the supply chain and awarding production to a new supplier is fraught with the expense and risk of moving operations and revalidating processes. Keeping production at Phase 2 would actually be the best business move, if it could meet the global price, efficiently expand capacity to handle the volume from Portsmouth, and stay profitable.
“When we were looking at all the alternatives, which we are obligated to do, most of what we looked at was outside the U.S,” said Mark Johnson, vice president of operations for Medtronic’s Surgical Technologies Business Unit, who would decide where to locate the business. He noted that in the medical device industry, it’s usually better not to move “if we can get a supplier to improve internal processes to give us improved quality, costs, and delivery.”
Phase 2 agreed to meet the global price. But the company’s all-in commitment to dramatically improve the performance of its lean operating system was another big factor in its keeping the business. That meant work would change for everyone, including staff and management. They would have to learn new behaviors for sustaining the new lean system and culture.
“By partnering in a lean journey, we felt that we could get a transformational change done across an entire company that also is a very, very important supplier for Medtronic,” Johnson explained. “We didn’t feel like we could do that with any of the options of sending work outside the U.S.”
Having margins trimmed to match the global price was an “eye opener,” said Prime. “That made us realize we had to cut costs and do things in a better [lean] system.”
Critically, the improved system introduced a new tool to support the technical changes and cultural development. Called a kamishibai board after a type of Japanese visual storytelling, it combined standardized work, accountability, and visual control into a tangible “golden triangle” of sustainability and culture change.
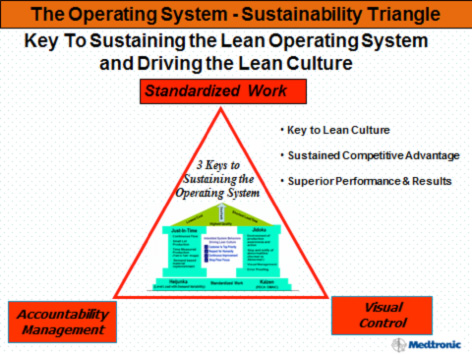 |
| The familiar lean production system house at Phase 2 is inside a “golden triangle” of sustainability. |
A Two–Part Change Strategy
First, the operating system had to improve. Prime and Troy Dayon, MAE operations director, directed the joint effort. Key lean improvement team members included Rick Henderson, Phase 2’s production and continuous improvement manager, and David Errico, lean deployment project manager for Medtronic’s Surgical Technologies Business Unit. Troy and Adam produced a two-part improvement strategy:
- Get material costs down immediately to close the gap between current and global pricing. Brenda Conkel, MAE senior engineering manager, led the effort while both companies worked together to make cost-savings improvements at the product component level and obtain price reductions from suppliers.
- Develop a lean Cell Operating System so Phase 2 could handle a three-fold growth in consolidated volumes with minimal growth in operating costs. Errico, who spent three years with Toyota’s Operations Management and Development Division, a special unit that helps implement the Toyota Production System in company facilities, guided this effort.
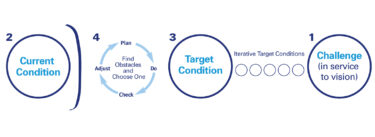
(For more details about the transformation approach Errico used, read the section beginning with “Lots of Lots” in the lean case study “Using Plan-Do-Check-Act as a Strategy and Tactic for Helping Suppliers Improve”)
The improvement team implemented the new system in two steps:
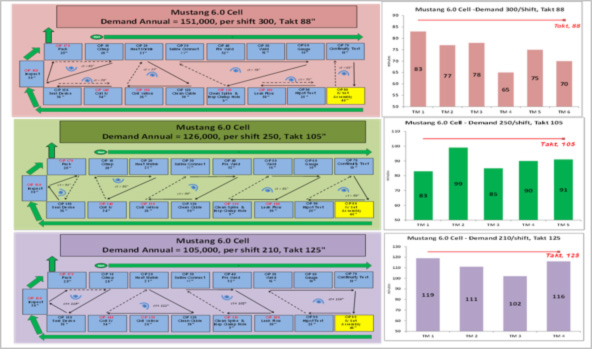
First, it created a U-shaped, single-piece flow cell for a high-volume product line making one type of medical device that would zoom in production volume from 60,000 units to 150,000 in a year. Cell targets called for a 33% improvement in productivity (measured as parts per person) and eliminating two production days of kitted components from inventory. To reduce inventory and drive up turns, the team developed an on-site warehouse to resupply the cell hourly based on kanban triggers.
The team designed the high-volume cell to respond to demand fluctuations by increasing or decreasing the number of operators based on three different takt times. A chart illustrating the takt times and operator walk paths for different staffing levels hangs at the cell:
 |
In the second implementation step, the team used lessons learned on the high-volume line to create a U-shaped, high-variety, single-piece flow cell to make nine types of medical devices.
Philosophy Lesson
The first lesson for Phase 2 in improving the high-volume line — and the overall operating and management systems — was a philosophy lesson, a way of thinking that Errico had experienced at Toyota. Every improvement project, he explained to managers and employees, would be guided by four values:
- The customer is our top priority
- Respect for humanity
- Continuous improvement
- Shop-floor focus
After analyzing the current state in assembly and an existing on-site warehouse for a month, Errico and the Phase 2 team found these key conditions:
- Daily demand fluctuated from a few dozen units to several hundred.
- To provide traceability of parts, a medical device requirement, components for 560 devices were picked from the warehouse, kitted in plastic bags, and staged two days before production as a “mother kit” outside the clean room production area.
- Team members seated at stations assembled devices in small lots of 10.
- After part identification data for each small lot was verified, the operator moved the small lot to the next station.
- Operators performed a lot of noncyclical work because the “mother kit” of 560 units was broken down into smaller quantities when it moved to production, leading to the need for operators to refill parts, complete paperwork, etc.
- The small lots were individually sealed in sterile barrier packaging, then aggregated for final inspection and final packing in a room outside the clean room, a step that added up to 10 hours to lead time.
The team decided the future- state process would have to follow this basic lean thinking:
- Run production to a level schedule.
- Develop real-time problem solving.
- Use a kanban system and a material handler to deliver components hourly to operators.
- Go from small lots to single-piece flow.
- Develop cells with visual management and flexible staffing to respond to different takt times when demand fluctuated.
- Sustain the new operating system and culture with a kamishibai system.
Killing Kitting
A big barrier to implementing this thinking was kitting. “If we could stop kitting, we not only eliminate wasted processing and non-value added labor,” said Errico, “we could go to single piece flow. If we go to single piece flow, our visual management gets down to a 12-minute pitch with an hourly status review, allowing us to see how effective our operation is, improve quality, and create an environment that drives on-the-spot improvement”.
Fortunately, the company had just upgraded its MRP system. Now it could allocate components to an order by scanning it into MRP. Every hour, MRP back flushed the parts from inventory.
The improvement preserved part traceability while permitting components to be stored at point-of-use. Parts were replenished hourly by a material handler using a tugger and a kanban system to retrieve parts from the redesigned warehouse, where a plan for every part (PFEP) set min-max levels for component flow racks. Parts were racked for easy accessibility. Material velocity increased without stock outs.
As planned, production was organized into a U-shaped cell and rebalanced for three different takt times and staffing levels to absorb fluctuations in demand. Each takt time had a different set of standard walk patterns within the cell that operators learned. “We called it dancing with the stars,” said Henderson about the training.
Because work stations in the cell were arranged inside a narrow U-shape, operators easily crossed from one side of the cell to the other during a walk pattern. The sequence of work elements operators performed could be different than the processing sequence.
One-piece flow through the cell removed much of the operators’ noncyclical work, which was absorbed by a material handler and team leader activities. “Now, with parts delivered to them, operators focused only on the value-added work,” said Errico.
Sustaining the New Lean System and Culture
Sustaining and building on one-piece flow and other improvements was aided by a kamishibai system. Developed by Toyota, kamishibai facilitates sustaining a lean operating system and culture. It uses a status board, observation cards, and a schedule for when leaders and designated staff members must visit production to observe an operation within a process. Thus, it firmly establishes the concept of “go and see” (genchi gembutsu) what’s really happening as an important part of management’s standardized work.
 |
| The high-volume cell kamishibai board with rack of observation cards is posted in the production clean room at Phase 2.
|
 |
|
Boxes on cards tell the observer what cell and operation to check, depending on which of three possible takt times are in effect. |
“It’s the engine for driving culture change because everybody in the business really serves the shop floor,” said Johnson.
The system has two elements: Kamishibai cards are small cue cards bearing process or system observation assignments for observers. At Phase 2, observers select assignments at random. This way, people know an observer will check an operation hourly, but no one knows which operation. All operations are evaluated every two shifts.
The kamishibai board lists what activities to check hourly. At Phase 2 that includes:
- Variance from hourly production targets written on the production white board.
- Adherence to the standardized work sheets hanging at each cell work station.
- Adherence to the correct walk path for the takt time in effect.
- If the countermeasure plan requires the observer to take any actions.
- If there are rejects in the observed operation’s red bin.
The observer initials the board and indicates the status of each activity with symbols:
Green circle = Normal condition
Yellow triangle = Abnormal condition with “contain and correct” action happening
X = Abnormal condition
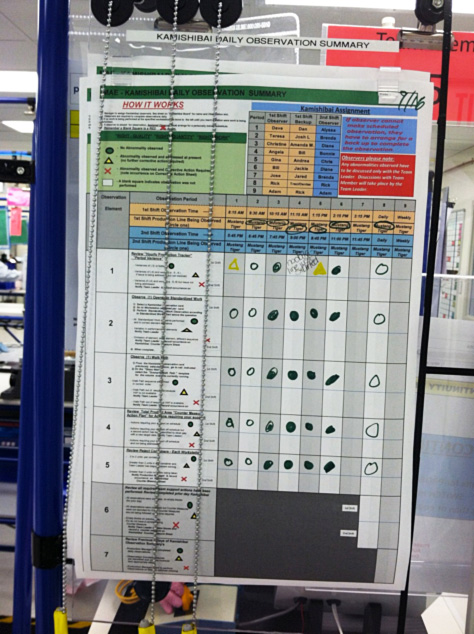 |
| A close-up of the high-volume board shows instructions and operation status symbols at the top left. The beige column at the top center lists the observers for each of two shifts through nine time periods. |
Staff members from support areas, such as quality or finance, and selected managers perform observations to identify abnormal conditions in key processes and systems. Each member or manager performs a 15-minute standardized observation at a workstation identified on their selected card. Observations happen every hour at defined times over nine periods while production is running.
Abnormal conditions are addressed immediately by team leaders and the affected operator with on-the-spot kaizen activity. Issues requiring additional follow-up or deeper investigation are documented and tracked on a countermeasure tracking system located at the cell. Any abnormalities not being addressed by countermeasures are elevated for attention, not only for the performance abnormality but also for failure to support the lean culture.
“The kamishibai changed our culture because everyone now is involved in supporting production,” said Henderson.
“It’s that but it also enforces [improvement] events so there is no backsliding,” said Prime. As president, he has the most work to do in the kamishibai system. Once per week, he makes the same standardized observations as his staff, reviews the previous week’s kamishibai charts, checks that the countermeasure system is being used and the countermeasure items are being addressed. As production manager, Henderson does a daily observation and does an overall check at the end of the shift.
“Kamishibai is a phenomenal tool, and I had never heard of it,” Prime said. “It was a game changer for us.”
Assessing the Different Lean Approach
The change isn’t lost on employees who see staff and managers using the board every hour to observe a process, Henderson noted. “They see everyone is engaged. It’s a missing link. It really is.”
He said establishing standardized work and tracking hourly output are insufficient to sustain a lean production system and culture. “If no one is overseeing it, you get busy with other work and soon no one is following standardized work, no one is fixing the to-dos. The operators figure no one cares.”
Errico said the system inverts the organizational pyramid. “The president of the company is now serving team members and has the most work to do on the kamishibai, followed by the production manager,” he explained. The entire management team is fully engaged in supporting the production team, all driving to the same goals.”
“What Dave taught us was an entirely different approach to lean that accelerated our journey by years,” said Prime. “It was more conceptual and philosophy based than just tool based. When you start with the right foundation and the right thought process behind any improvement event, you wind up with a much deeper understanding of what the true intent is, not just how to do 5S but why you are doing 5S and how it rolls into the whole process. It brought another layer for us and it’s helped us tremendously.”
Said Johnson, the Medtronic VP, “I absolutely think this is a great example of a small company doing something they didn’t think they could do. In the end, we got the financial benefit we needed and we didn’t have to send work outside the Unites States. It was a win, win, win on all sides.”
Here’s an overview of the transformation in pictures from the shop floor:
 |
 |
 |
|||
|
Assembly work was redesigned … |
Into flexible U-shaped cells with one-piece flow… |
With a system of standardized work …
|
|||
 |
 |
 |
|||
|
That included heijunka devices to level production. (At the start of each shift, team leaders load the rack with an hour of work. Each card is one device and each slot is a 12-minute pitch.) |
The plan-for-every-part sets standards for warehouse locations and quantities of every part used in production. | Phase 2 President Adam Prime, left, shows a visitor a wall displaying key metrics in standard fashion for production and support areas. | |||
|
|
|||
|
|
Before the changes |
After |
|
|
|
|
|
|
|
High-volume annual volume |
50,000 units |
150,000 units |
|
|
Productivity |
33.3 parts/person/shift |
50 |
|
|
High-volume cell floor space |
363 sq. ft. |
198 sq. ft. |
|
|
Work in Process |
360 units |
86 units |
|
|
Pack out cell |
1 day |
¼ day |
|
|
First run yield |
91.3% |
98% |
|
|
Warehouse materials on hand |
50 days |
25 |
|
|
Kitted parts on hand |
2 days |
0 |
|
|
|
|
|
|
Relevant links for learning more:
- Phase 2
- Read a followup story to the case study: Manufacturing Balancing Act: Pull Versus ERP
- Medtronic Advanced Energy
- The Lean Enterprise Institute
- Read the related Lean management case study: Using Plan-Do-Check-Act as a Strategy and Tactic for Helping Suppliers Improve
- Join LEI’s Lean Community for access to case studies, webinars, newsletters, lean management columns, e-letters, and many other resources for starting and sustaining a lean transformation.